A method for separating zinc from an industry product using a pyrometallurgical reaction
Pyrometallurgical route for selective zinc separation: reduced metal reacts with oxidised zinc at a controlled interface under non-oxidising gas, enabling volatilisation and removal of zinc from complex residues.
- Jurisdiction
- WIPO (WO)
- Publication
- WO2022037781A1
- Published
- February 24, 2022
A Carbon-Free, Multi-Metal Pyrometallurgical Process for Direct Metallic Zinc Recovery from Electric Arc Furnace Dust
Introduction
The primary use of zinc today is for galvanizing steel, which protects the steel from corrosion. At the end of its lifecycle, galvanized steel becomes scrap, and electric arc furnaces recycle this scrap into new steel products. This recycling produces a byproduct called electric arc furnace dust (EAFD). EAFD is classified as hazardous waste, yet it remains the largest secondary source of zinc currently available. Besides zinc, it contains significant amounts of other valuable metals—particularly copper, nickel, and tin—which, in today’s recycling methods, typically end up in landfills and are permanently lost. Efficient recovery of these metals from EAFD is therefore crucial for achieving a truly circular economy.
In Europe alone, processing EAFD via the traditional Waelz kiln method leads annually to approximately 25,000 tons of zinc, 2,000 tons of copper, and 800 tons of nickel lost through Waelz slag disposal. Furthermore, this approach results in roughly 500,000 tons of CO₂ emissions each year. These substantial environmental and economic losses clearly show the need for cleaner and more resource-efficient recycling processes.
Conventional zinc recovery in the Waelz kiln involves carbothermic reduction of zinc oxide (ZnO), which produces gaseous metallic zinc. However, due to the presence of CO₂ and H₂O, the gaseous zinc reoxidizes immediately to zinc oxide particles:
ZnO(s) + C(s) → Zn(g) + CO(g)
Zn(g) + 1/2 O₂(g) → ZnO(s) (highly exothermic, heat recovered)
This reoxidation reaction is highly exothermic, and the energy released is recovered in Waelz processes. Any competitive alternative process would either need to recover this energy or—ideally—prevent reoxidation altogether.
Our patent application describes a fundamentally different, carbon-free pyrometallurgical approach based on recent kinetic discoveries. Specifically, instead of relying on carbon, our process leverages a metallothermic reduction between zinc oxide and metallic iron:
ZnO(s) + Fe(l) → Zn(g) + FeO(s)
This metallothermic pathway produces only metallic zinc vapor without oxidizing gases (CO₂/H₂O), thereby completely avoiding reoxidation. The absence of reoxidation eliminates the need for heat recovery from the exothermic oxidation step entirely, as the step simply does not occur. Additionally, the direct condensation of metallic zinc is technically straightforward, producing metallic zinc directly instead of intermediate zinc oxide concentrates.
Furthermore, our metallothermic concept inherently facilitates multi-metal recovery. By selectively oxidizing iron, it allows copper, nickel, and tin to accumulate as a valuable alloy. This turns traditionally low-value scrap into an economically attractive resource, transforming the process from simple zinc recycling into a genuine multi-metal recycling method.
Fundamental Scientific Principles
Zinc recovery from electric arc furnace dust traditionally occurs in a two-step reduction and reoxidation cycle. First, zinc oxide (ZnO) in the dust is reduced by carbon, forming gaseous metallic zinc along with gaseous by-products such as CO₂ and H₂O. Immediately afterward, due to these oxidizing gases, the zinc vapor reoxidizes into finely dispersed zinc oxide particles. These particles are then separated from the furnace off-gas stream using baghouse filters, producing a high zinc oxide concentrate called Waelz oxide. Waelz oxide must subsequently undergo the complete primary zinc production route, making the process energy-intensive and complex. Direct condensation of metallic zinc vapor is practically impossible under these conditions, as even trace amounts of CO₂ or H₂O cause rapid reoxidation.
In contrast, metallothermic reduction using metallic iron eliminates the formation of oxidizing gases entirely. As a result, metallic zinc vapor remains stable and can be condensed directly into metallic zinc without risk of reoxidation. This simplifies the zinc recovery significantly and enables the direct use of metallic zinc—for example, in galvanizing steel—or allows straightforward refining, if necessary.
Kinetic Modelling as Basis for Process Innovation
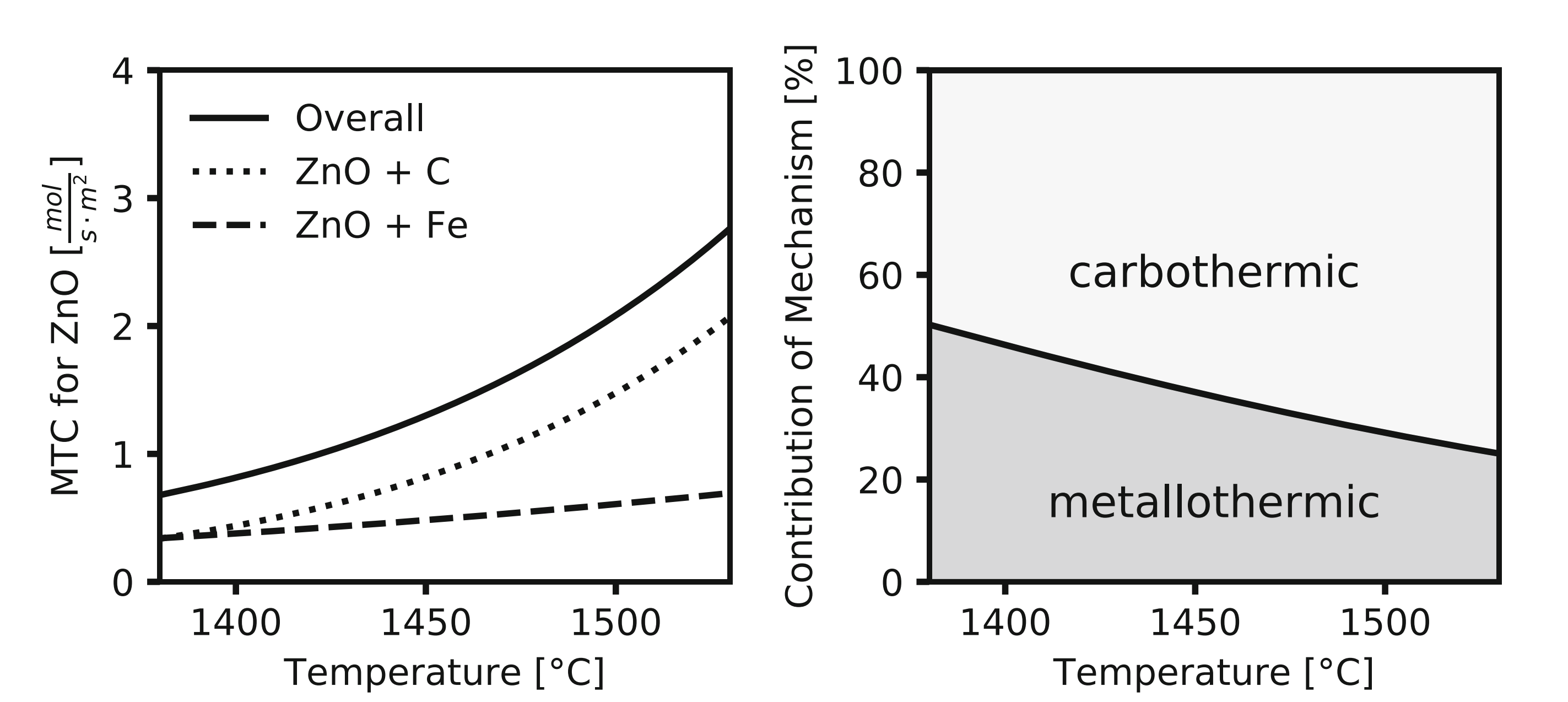
This innovation was developed following detailed kinetic studies on zinc oxide reduction using carbon-saturated iron baths. Initially, the reduction was intended to be carbothermic, using the carbon dissolved in iron. However, kinetic modeling revealed an unexpected result: at higher temperatures, the reaction between metallic iron and zinc oxide (metallothermic reduction) dominated the overall reduction process (Leuchtenmüller, 2021; 2022). This discovery formed the foundation for developing a completely carbon-free recovery method.
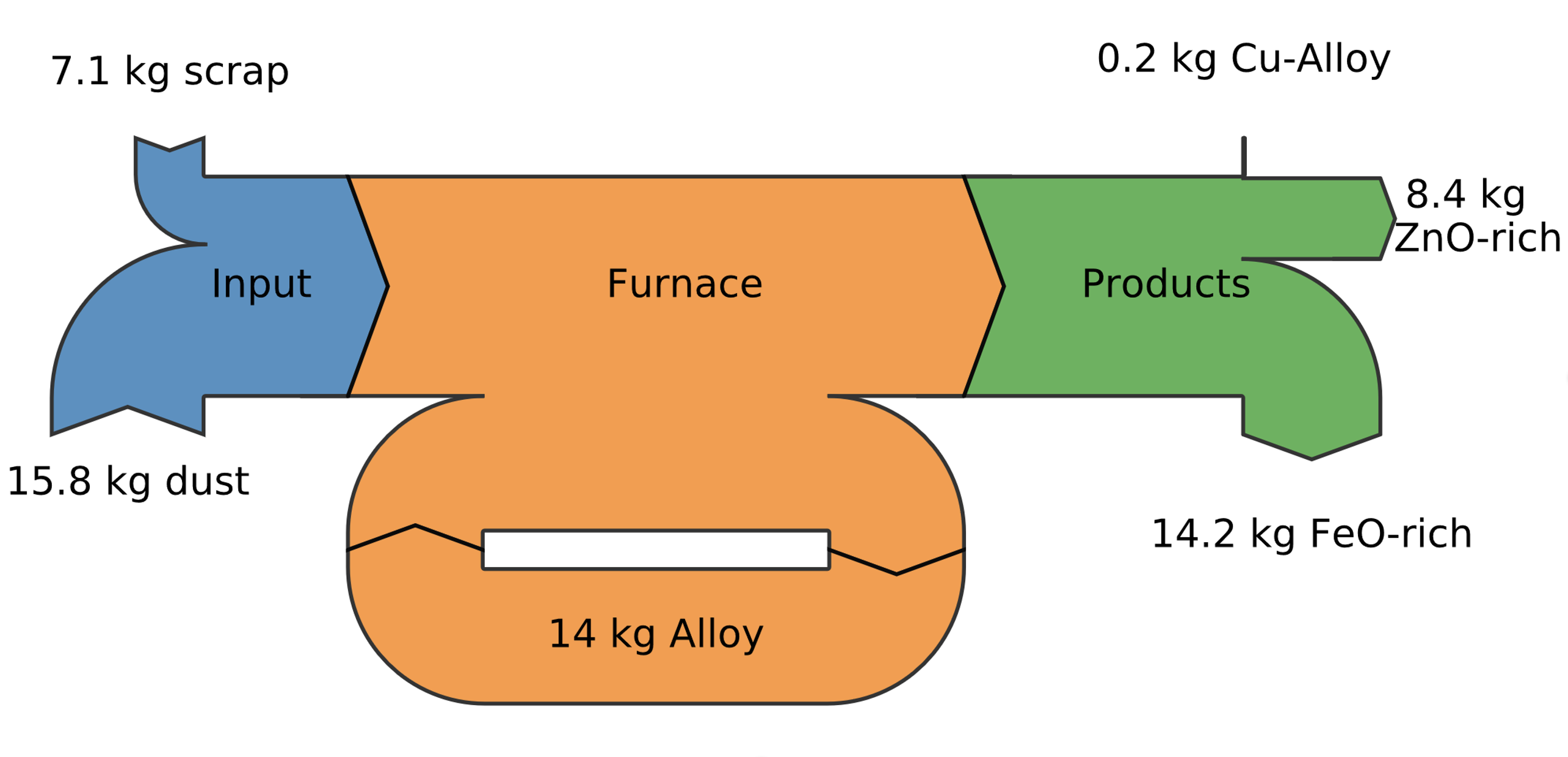
The resulting novel pyrometallurgical process leverages this rapid metallothermic reaction to directly produce metallic zinc from low-grade scrap materials, including those with elevated copper content. Due to the controlled oxidation of iron, more noble elements such as copper, nickel, and tin accumulate in the metal bath, forming a valuable alloy. This alloy can subsequently be sold to e.g. copper smelters for further refining in converters or anode furnaces. Thus, this new recycling method represents the first practical concept for efficiently recovering copper and other valuable metals directly from low-grade iron scrap.

Environmental and Economic Evaluation
Using low-quality scrap materials containing higher levels of copper significantly reduces the environmental footprint of zinc recycling. This metallothermic approach eliminates the CO2 emissions typically associated with traditional EAF dust recycling, as it avoids carbon entirely. Additionally, by recovering metallic zinc directly, this method removes the need for the costly and energy-intensive refining processes required to convert traditional Waelz oxide into metallic zinc.
Importantly, the simultaneous recovery of a copper-enriched alloy containing nickel and tin transforms what was previously a single-metal recovery operation into genuine multi-metal recycling. Low-grade scrap, typically of limited economic value, now becomes a valuable resource contributing directly to industrial metal supplies.
Finally, the iron oxide-rich slag produced by this process can serve as a high-quality secondary raw material for ironmaking. Because this slag is free from volatile elements and copper, it is particularly suitable for efficient use in advanced counter-current reduction reactors, further enhancing resource efficiency and sustainability.